Democratization and accessibility of complex engineering tools
Our mission is to develop advanced engineering tools that are both affordable and accessible to non-engineering laboratories. In collaboration with experts in developmental biology, the microbiome, infectious diseases, and clinical research, we have created a suite of innovative technologies designed to enhance the study of human physiology and disease. Use the links below to request more information or explore potential collaborations.
-
We built an organ-on-chip for less than $10. Now we need help putting it in every lab that needs one.
Infectious disease researchers studying STIs. Maternal health scientists modeling preterm birth. Kidney biologists investigating developmental disorders. Cancer researchers screening drugs against tumor microenvironments. Immunologists studying gut inflammation. Lung disease researchers. Pharmacologists testing therapeutic candidates.
All of them need the same thing: human-relevant tissue models they can build and run in their own labs. Organ-on-chip technology, which is miniaturized living tissue models built from human cells, can model organ function more accurately than cells in a dish and more faithfully than animal models. Better disease modeling. Faster drug screening. Fewer animals. Applicable across virtually every organ system and disease area.
But most platforms cost $100–$400 per chip, require cleanroom fabrication, and demand engineering expertise that most biology labs don’t have. The researchers closest to the diseases, microbiologists, immunologists, clinicians, etc., are locked out of the tools they need most.
We built something different.
Less Than $10. No Cleanroom. It Works.
Our platform costs less than $10 per chip and can get significantly cheaper at scale. It uses commodity materials, standard lab equipment, and a modular design that any biologist can learn. No cleanroom. No microfabrication. No engineering degree required.
The design is intentional. We built this by researchers, for researchers to be compatible with the equipment and methods already in a biology lab, specifically to lower the barrier to entry and amplify success. Swap insert types, channel geometries, or cell configurations without redesigning the entire device. Go from a completely new design to a working chip in one hour. Build your tissue model under static conditions you’re comfortable with, then connect to flow when ready.
One platform. One set of methods. Cervix, kidney, lung, gut, placenta, lymph node; any organ system.
The Technology Problem Is Solved. The Distribution Problem Isn’t.
We’ve transferred this platform to 14 independent labs across 6 countries on 4 continents, all non-engineering biology labs that had never built a microfluidic device, and some that have never used a pump. They got it working through our validated protocols and instructional videos.
In our most rigorous test, independent labs built cervical tissue models and demonstrated complete Chlamydia and Gonorrhea infection cycles, microbiome-mediated immune protection, and immune cell recruitment; all on-chip, all validated across multiple labs. Published in Science Advances.
Researchers keep contacting us. They want to study cervical ripening in pregnancy, the role of the microbiome in preterm birth, HPV pathogenesis, kidney development, gut inflammation, and lung disease. The demand is real and growing faster than we can support it.
This is what happens when you put accessible tools in the hands of domain experts. The questions and the discoveries follow.
Where This Is Going
We are actively expanding the platform:
Stromal tissue integration — adding embedded stromal cells and tissue-resident immune cells, currently in trials at partner labs
New organ systems — adapting our previous placenta and lung models, building out oral, gut, and lymph node tissues
Parallelization and real-time sampling — expanding capabilities for higher-throughput screening
Training infrastructure — bootcamps, on-site deployment, and visiting researcher programs, so labs can get operational fast
Every experiment run on a human-relevant tissue model is one that doesn’t require an animal. The FDA and many countries internationally are actively moving away from mandatory animal testing toward organ-chip approaches. For that regulatory shift to matter, these tools can’t remain in a handful of engineering labs or commercial vendors priced for regulatory screening. They need to be everywhere.
The Vision: Scientific Infrastructure, Not a Product
We want to build a network of regional manufacturing and training hubs. Any location with a couple of benchtop hobbyist tools becomes a node. As we and others using the platform validate new organ system designs, they push through the network. Each trained lab can train its neighbors. Shared protocols, reproducible science, and rapid advancement of open-source science. The platform becomes shared infrastructure for a new way of doing biology.
There are tens of thousands of biology labs worldwide that could use human tissue models but can’t access them today. Problems that are regional or global in scope, from neglected tropical diseases to preterm birth, become tractable when the researchers closest to them can build and access the models they need. New therapeutic targets emerge. New screening approaches become possible. The pace of discovery accelerates.
We’ve shown that the technology works. We’ve shown it transfers. We’ve shown what becomes possible when domain experts have the tools.
Now we need philanthropic support to scale it.
We are seeking foundation partnerships and philanthropic donations to put this platform in hundreds of labs worldwide — to fund device fabrication, starter kits, platform adaptation for new organ systems, training programs, distributed device making nodes, and the staff to support technology transfer at the pace that demand requires.
The tools exist. The evidence is published. The demand is real. What we need now is the support to scale.
Other Models

Parallel Plate Flow Chamber
-
Zonal patterning of extracellular matrix and stromal cell populations along a perfusable cellular microchannel, B Chernokal*, BJ Ferrick*, JP Gleghorn, Lab on a Chip, 2024, 24, 5238 - 5250. *indicates equal contribution
We have developed and validated a simple strategy to create an in vitro microphysiological system with regionally patterned stromal cell populations and hydrogel properties along the length of a perfused continuous tubular epithelium. This highly customizable system has wide utility for modeling epithelial and endothelial tissue interactions with heterogeneous hydrogel compositions and/or stromal cell populations to better dissect the specific microenvironmental factors that govern developmental tissue patterning.
Click here for more information or to inquire about a collaboration
In-house required equipment:
Incubator
Biosafety cabinet
Peristaltic pump with flow rate control down to at least 0.01 mL/min
-
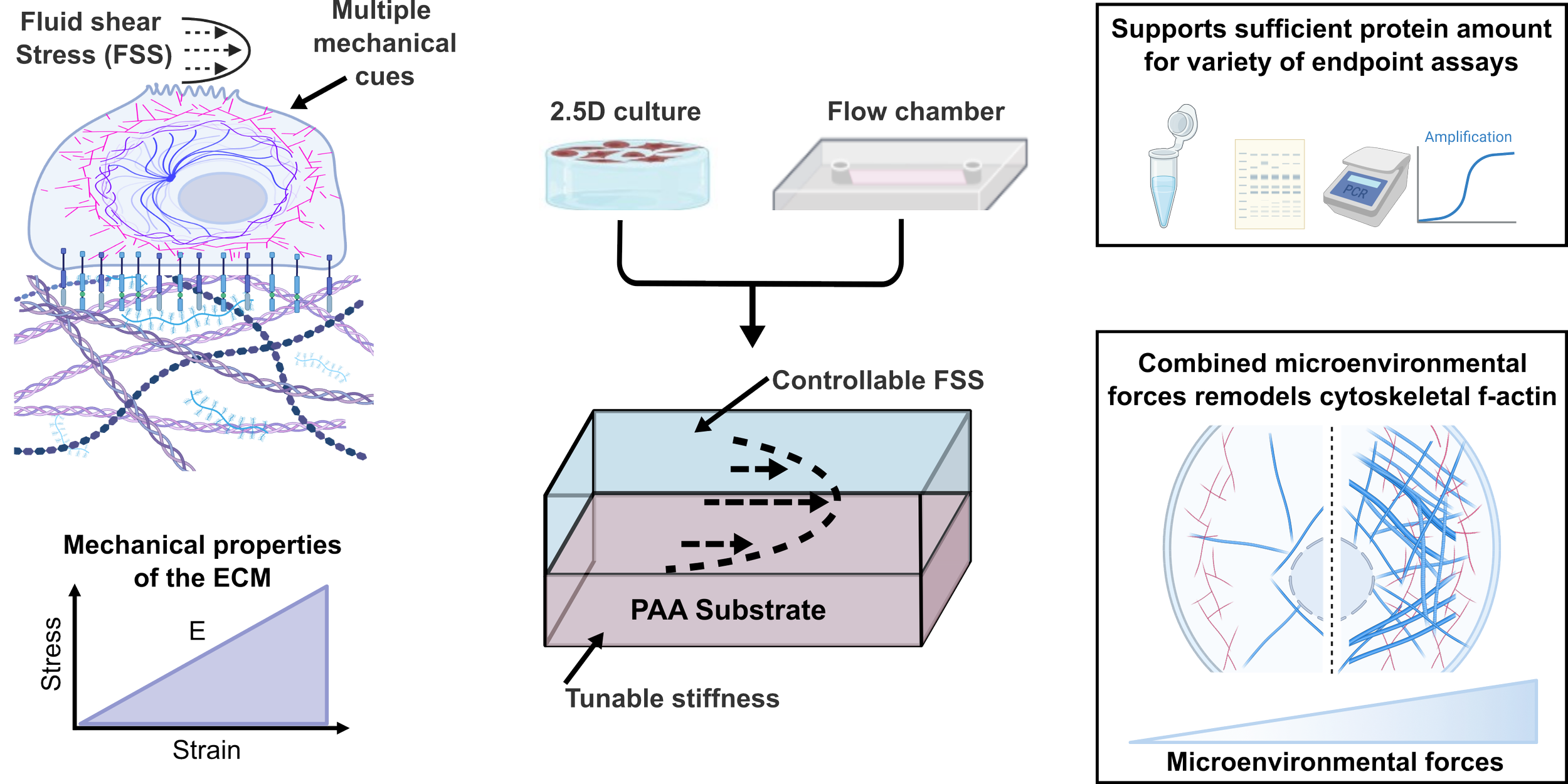
Modular parallel plate flow chamber with tunable substrate mechanics and defined shear stress, BJ Ferrick* & JP Gleghorn, Biomedical Microdevices, 28:8
In this work, we developed an accessible parallel-plate flow chamber to enable independent control of both extracellular matrix stiffness and fluid shear stress, two key mechanical cues that cells experience in their native environments. Our device maintains predictable fluid dynamics, supports robust epithelial monolayers, and provides enough material for downstream biochemical analyses, addressing major limitations of existing microfluidic systems. Using this platform, we discovered that stiffness and shear stress act synergistically to remodel the actin cytoskeleton, highlighting how cells integrate multiple mechanical signals.


Zonal Patterning Device
PMN (pink) migrating towards Ng(yellow) through an endocervical A2EN (cyan) cell layer in the device.
Click to start movie
Protocols
Resources
Large Format PAA gels
CS Millar-Haskell, JP Gleghorn. (2023) A large-format polyacrylamide gel with controllable matrix mechanics for mammalian cell culture and conditioned media production. Bio-protocol 13(17).
BEADS: Semi-automated 3D Fibrin Angiogenesis Workflow
Tissue sectioning and staining for 3D reconstruction
MA Kibler, MD Miller, MJ Donzanti, JP Gleghorn (2025) Confocal-Compatible Workflow for Sectioning, Staining, and Imaging Serial Vibratome Sections for 3D Anatomical Reconstruction of the Lymph Node, BioRxiv, 10.1101/2025.10.29.685350
CR Gonyea, B Hayward-Piatkovskyi, JP Gleghorn (2026) BEADS: An Interactive Semi-Automated Workflow for 3D Fibrin Angiogenesis Assays Enabling Co-Culture and Directionality Analysis, Microvascular Research, 165, 104915
Parallel Plate Flow Chamber part drawings
Modular Microphysiologic Device
Democratizing Organ-on-Chip Technologies with a Modular, Reusable, and Perfusion-Ready Microphysiological System, DJ Minahan*, KM Nelson*, F Ribeiro, BJ Ferrick, A Zurzolo, K Byers, and JP Gleghorn, Advanced Healthcare Materials, e02202, *indicates equal contribution
We have developed and validated a simple strategy to create a two layer in vitro microphysiological system within a one-time-use cell culture insert and a reusable cassette to enable fluid flow. The system enables easier cell culture than PDMS based devices and can be irreversibly sealed to enable access to the cells for assays. This highly customizable system has wide utility for modeling transport across a cell barrier and cell-cell interactions to better study physiological and pathogenic processes.
A Microphysiologic Model of the Cervical Epithelium Recapitulates Microbial, Immunologic, and Pathogenic Properties of Sexually Transmitted Infections, KM Nelson, DJ Minahan, VL Edwards, IJ GLomski, DJ Delgado Diaz, K Thomas, FC Walker, PM Bavoil, I Derre, AK Criss, J Ravel, & JP Gleghorn, Science Advances, In Press.
We introduce a validated microphysiologic model of the human cervix that recreates key features of the cervicovaginal environment, including epithelial barrier function, microbiome interactions, and host responses to infection. We demonstrate that the platform captures clinically relevant dynamics of sexually transmitted infections, providing a powerful tool to uncover mechanisms driving women’s reproductive health and disease. As an accessible and modular system, we designed this model to support broad adoption and welcome collaborations to expand its use across new areas of maternal and women’s health research.
Click here for more information or to inquire about a RESEARCH COLLABORATION
Click here to contact us to HELP DEMOCRATIZE these models
In-house required equipment:
Incubator (hypoxic preferred)
Biosafety cabinet
Multichannel peristaltic pump with flow rate control down to at least 0.01 mL/min